Maternity epilepsy toolkit updated Aug. 2024 to help guide women and their maternity and neurology teams with epilepsy management during pregnancy and postnatal.
For WORD version, contact Kim Morley @epilepsymidwife@gmail.com
Maternity epilepsy toolkit August 2024 p[...]
Adobe Acrobat document [562.0 KB]
Twitter @epilepsymidwife
This resource has been developed by an epilepsy specialist nurse/midwife from the experience of supporting women with epilepsy & healthcare professionals over the last two decades. It showcases the evidence based research & guidelines & links with epilepsy & pregnancy support groups. It is regularly updated to reflect opinions of women with epilepsy & health professionals.
Read the latest on epilepsy medicines in pregnancy
10/06/2025 Updated safety and educational materials from MHRA regarding valproate exposure in pregnancy and information about increased risk of low birth weight or small for gestational age with pregnancy exposure GOV.UK
05/09/2024 MHRA advises men and their partners taking valproate to use effective contraception. Please click onto link for latest guidance GOV.UK
20/06/24 New pregnancy prevention measures introduced for topiramate. Topiramate must no longer be prescribed to women and girls unless they fulfil the requirements of a Pregnancy Prevention Programme. GOV.UK
12/12/22 How effective are different form of contraception? NHS
Check your antiseizure medicine does not interact with hormonal contraceptives: Epilepsy Action Epilepsy Society
GOV.UK alert: women taking sodium valproate or valproic acid. The brand names are Epilim, Epilim Chrono, Epilim Chronosphere, Episenta, Epival, Depakote, Convulex, Kentlim, Syonell, Valpal & Belvo. Click Patient valproate booklet There is specific information for girls and young women taking sodium valproate, click: Medicines for children
Please obtain accurate information and advice from your GP, epilepsy specialist doctor or nurse before making any changes to your epilepsy medicines. This is important as untreated epilepsy can lead to increased risk of serious harm from uncontrolled seizures. To reduce unplanned pregnancy risk, click NHS guide link: contraception
Pre-pregnancy advice to prepare for pregnancy
Support
It is advisable that you receive support during the child-bearing years. It is important not to stop your epilepsy medication without seeking accurate advice from a doctor or epilepsy specialist due to risks of untreated epilepsy. If your diagnosis has not been reassessed in adult years, please discuss this with your GP before considering pregnancy.
It is important that you have the opportunity of having a medication review before pregnancy. However, the type of epilepsy can limit choice of treatment. Some epilepsies, particularly generalised (genetic) may only respond to valproate. All women should receive accurate information about the possible risk of epilepsy medication on a developing baby versus risk to the them of changing their medication.
Decision making
Many women with epilepsy are understandably anxious about pregnancy. To help with your decision making, you should be provided with accurate evidence based advice about fertility, contraception, pregnancy, breast feeding and safe parenting. Feel reassured that the majority of women with epilepsy have normal pregnancies and birth healthy babies especially when their pregnancy is planned.
Frequently asked questions
What is epilepsy?
Epilepsy is a serious neurological disease in which unprovoked, unpredictable seizures occur as a result of sudden bursts of excessive electrical activity in the brain.
The diagnosis should either be made by a specialist paediatrician in childhood or a medical practitioner also with training and expertise in epilepsy, in adulthood, following at least two unprovoked (or reflex) seizures occurring more than 24 hours apart; or following one unprovoked seizure and a probability of further seizures similar to the general risk of recurrence (at least 60%) after two unprovoked seizures occurring over the next two years; or a diagnosis of an epilepsy syndrome [1].
Epilepsy is usually diagnosed before age 20 and over age 65; but can occur at any time. The symptoms are individual to the person they are affecting and dependant on where in the brain they start, and where the abnormal electrical activity spreads within the brain. If a seizure starts in one part of the brain, it is called focal and if it involves most of the brain at the outset of the seizure, it is called generalised. Sometimes people can have a combination of seizure types or their epilepsy cannot be classified. Further information about what epilepsy is, how it is diagnosed and treated and information to help you take control of your condition, can be obtained from these excellent support groups: Epilepsy Action, Epilepsy Society, Epilepsy Research, SUDEP Action, Epilepsy Scotland
Learn more about epilepsy Epilepsy and you and for younger girls click: Me and My epilepsy: Muir Maxwell Epilepsy Centre
Are all seizures due to epilepsy?
No, seizures can be caused by a number of conditions including: syncope (fainting) due to a drop in blood pressure, lack of oxygen (anoxia) or a cardiac condition such as long QT wave. Syncope and non epileptic attack disorder characterised by disassociative seizures (also called non-epileptic seizures) are the leading causes of epilepsy misdiagnosis. If you have been diagnosed with a psychological cause for your seizures, do access excellent information and support from Non-epileptic attacks info: NEAD or Neurosymptoms.org. Please note that some people with known epilepsy can also experience non-epileptic seizures. Other causes of misdiagnosis include migraine, sleep disorders and paroxysmal movement disorders [2]
Metabolic disorders such as diabetes related to very low or high blood sugar; low sodium (salt); hypercalcaemia (high blood calcium levels) and thyroid dysfunction can provoke seizures. Many prescribed and unprescribed drugs, alcohol, some homeopathic drugs and recreational drugs can also lead to seizures.
Febrile convulsions (seizures usually related to raised temperature in the very young), stroke, head injury/trauma, meningitis, encephalitis, eclampsia, (a rare but serious cause of seizures in pregnancy), brain tumour, cerebral malaria, electric shock [3] and toxic shock syndrome are examples of conditions that can present with convulsive or non-convulsive seizures. This does not necessarily mean the person will go onto develop epilepsy however; having a history of any of these causal conditions can significantly increase the risk of developing epilepsy.
Do many women with epilepsy have babies?
Yes; about 2,500 births occur annually to women with epilepsy in the UK.
Should I be referred for pre-conception counselling?
The majority of women treated with epilepsy medications have good seizure control. However, it is important the GP refers you to a specialist if your epilepsy diagnosis & treatment has not been reassessed recently by an expert medical practitioner in epilepsy; if you are taking sodium valproate; if you have uncontrolled seizures despite taking medication; if you are taking more than one epilepsy medicine; if you have nocturnal seizures; if you have a history of prolonged seizures or if you have been more than two years seizure free whilst taking epilepsy medication. If you do not meet any of these criteria, you can still ask for an expert opinion. The specialist will provide advice to enable you to make a shared informed decision about your future epilepsy management in preparation of pregnancy & parenting.
Why should I plan my pregnancy?
The majority of women have a healthy baby following a normal pregnancy and labour but it is important to reduce the risk of harm through careful pregnancy planning. Epilepsy medications can be associated with an increased risk of birth defects. The risk of these problems is often low but will depend on the type and number of epilepsy medications prescribed; the dosage, family history of congenital malformations and other conditions such as thyroid disease and diabetes. A safety leaflet on epilepsy medicines and pregnancy to help patients and their families understand the risks, click GOV.UK
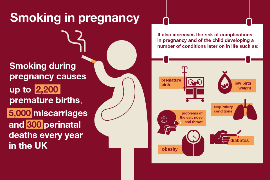
Alcohol [4.5] and smoking [6] are also linked to an increased risk of congenital malformations and other complications in pregnancy. The risk of congenital malformations and a fetal anticonvulsant syndrome which can include developmental problems and intellectual disabilites, is particularly increased in women prescribed sodium valproate in pregnancy. Please click on GOV.UK Patient valproate booklet for further information if you are taking this medicine. The other names for sodium valproate and valproic acid are: Epilim, Epilim Chrono, Epilim Chronosphere, Episenta, Epival, Orlept, Syonell, Valpal, Kentlim, Depakote and Convulex. For any women or girl taking valproate Click: GOV.UK for temporary advice during coronavirus (COVID-19) about the valproate pregnancy prevention programme. If there is risk of pregnancy, it is recommended girls/women use long acting reversible contraception: click FPA Patient guide. Check with your doctor that none of the other medications you take interact with the contraceptive implant. A guide for your doctor click: CEU Clinical Guidance: Drug interactions with hormonal contraception
The UK Teratology information service website is a resource for medicines safety advice in pregnancy/breast feeding. Information includes advice about: sodium valproate, topiramate, gabapentin, carbamazepine, pregabalin, gabapentin and lamotrigine. Click: search engine for further details on the UKTIS website; please note their Terms and conditions and privacy policy as information may not necessarily be current. If you are unable to obtain accurate information, contact the UK Epilepsy & Pregnancy Register for accurate information about potential risk of your particular epilepsy medication regime.
Can I stop taking my antiseizure medicines?
Do not suddenly stop your epilepsy medication without seeking urgent medical advice from your GP/epilepsy specialist/neurologist. Suddenly stopping epilepsy medication or missing doses can be associated with uncontrolled seizures resulting in serious harm and in rare cases, sudden unexpected death in epilepsy. This risk is increased in pregnancy.
Asses your medicines behaviour
How many doses of your medicines have you missed in the last 2 weeks?
What seems to get in the way of you taking your medicine?
What was going on when you missed a dose this past week?
When are you most successful at taking your medicine according to plan?
What can help me take my medicine?
- Understand when you should be taking your medicines. Ask your pharmacist to confirm the dose and timing of each medicine you are taking. Take the exact dose prescribed.
- Check the patient information leaflet inside the medicines box to see if you should take your medicine before or after food.
- Remember to put in for a repeat prescription at least 7 days before your medicines is due to run out.
- Write or electronically record your daily medication routine on a calendar or chart. Printable medication schedule templates are available online. Update your schedule if your medicine or dosage is changed.
- Keep your medicine schedule in an obvious place like the fridge door or a bedroom or kitchen cabinet.
- Keep your medicines in a safe, dry, cool place out of the reach of children.
- Ask your pharmacist for help if you do not understand your medicine plan.
- Consider using a daily or weekly pill organiser.
Is contraception affected by my epilepsy medication?
Some epilepsy medications induce enzymes in the liver which reduce the effectiveness or oral contraceptives, the implant, emergency contraception, hormonal patches and the reliability of natural methods of contraception. In addition, the combined oral contraceptive can reduce the effectiveness of lamotrigine. Therefore, it is strongly recommended that you seek advice from your GP or contraception and sexual health clinic in advance of sexual activity to reduce the risk of unplanned pregnancy. Click on: Care of baby & you for more detailed contraception advice and Epilepsy Action contraception advice. Advice for your professional from: Drug interactions with hormonal contraception, Click: CEU Clinical Guidance and Multi-drug Interaction Checker Medscape.com drug/drug/interaction/checker
Do I need to take folic acid?
It is currently recommended in national guidelines in the UK that if you take epilepsy medication that your GP prescribes folic acid 5 milligrams once daily for three months before you stop contraception or if there is risk of unplanned pregnancy. The advice recommends that you continue this until you are 12 weeks pregnant as it is thought to reduce the risk of neural tube defects, including spinal bifida [7]. This dosage of folic acid cannot be bought over the counter; it has to be prescribed by your GP who will ensure this is a safe drug and dosage for you to take with your epilepsy medication regime. There is data that folic acid taken in pregnancy may reduce the risk of a child having autism in women taking epilepsy medications however, further research is required in this area [8]. Further research is also required into what is the safest dose of folic acid to recommend women with epilepsy take during pregnancy and whether the higher prescribed dose is safe and offers any extra protection [9,10].
What can I do before pregnancy to reduce risks?
Please click on links for further advice and risk reduction strategies
- Smoking can be associated with increased risks to you and your developing baby; do take advantage of the NHS support to quit smoking NHS advice
- Achieve a normal weight and adopt a healthy lifestyle NHS Pre-conception care
- Stop drinking alcohol and seek advice from your GP about other prescribed and unprescribed medicines/substances used. NHS Support to quit alcohol
- Seek advice from your GP if you have mental health problems or any other health condition that may deteriorate during pregnancy. Mental health problems in pregnancy
- Inform the DVLA if you have a seizure/if you are doing an epilepsy medication change DVLA epilepsy and driving
- Take control of your epilepsy through increasing your knowledge by learning as much about it as possible. Epilepsy Action Epilepsy Society Epilepsy Research.
- Encourage your family and wider support network to increase their knowledge Epilepsy Action Advice for seizure management.
- Become an expert in managing your condition through Epilepsy Action epilepsy and wellbeing
- Keep a seizure diary and if possible and safe to do so, ask a family member to video one of your seizures. This helps specialists classify your seizure type, guide your treatment and advise you about safety. Epilepsy Society seizure diary advice Epilepsy Action seizure diary
- Wear an alert bracelet or ID jewellery. Lots of great designs available; please look up on your search engine.
- Reduce your risks SUDEP Action Epilepsy self-monitor Epilepsy Society: Risk assessment advice
- Always take extreme caution around water: showering instead of bathing when alone or when no one is around to ensure your well-being; the same applies to a jacuzzi or Hot Tub. Always ensure a pool lifeguard is aware of your epilepsy and swim with a buddy. Avoid being close to any water's edge alone.
- Ask your GP for a full blood screen including: Full blood count, Liver, renal, bone and thyroid function. A vitamin D level and ferratin level. If you take epilepsy medication, a baseline pre-conception serum level can provide guidance if your specialist team and you would like a combination of clinical and therapeutic drug monitoring in pregnancy.
- Improve your pre-conception health Emma's Diary Getting pregnant & having a baby
- Evidence based resources for expectant parents www.babycentre.co.uk
- Important for women to optimise their bone health: Royal Osteoporosis Society: Bone health advice
References & further reading
1. Fisher RS, Acevedo C, Arzimanoglou A et al (2014) A practical clinical definition of epilepsy. ILAE Official Report. Epilepsia 55(4): 475-482
2. Hernandez-Frau PE and Benbadis SR (2011) Pearls & oy-sters: errors in EEG interpretations: what is misinterpreted besides sharp temporal transients? Neurology 76: 57-e59.
3. Grell K, Meersohn A, Schüz J and Johansen C (2012) 'Risk of neurological diseases among survivors of electric shocks: A nationwide cohort study, Denmark, 1968-2008', Bioelectromagnetics 33(6): 459-465
4. Lacey J (2016) 'Reducing alcohol harm: early intervention and prevention', Community Practitioner 89(2): 26
5. Callanan C (2013) ''Binge drinking' among mothers raises number of children with fetal alcohol spectrum disorder', Learning Disability Practice 16(2): 6-7
6. Cope G (2015) 'How smoking during pregnancy affects the mother and fetus', Nurse Prescribing 13(6): 282-286
7. NHS Choices (2016) Why do I need folic acid in pregnancy? Health questions-NHS Available from: http://www.nhs.uk/chq/pages/913.aspx?categoryid=54
8. European Academy of Neurology. "Antiepileptic drugs during pregnancy: Folic acid could help to prevent autism." ScienceDaily. 31 May 2016
9. Asadi-Pooya AA (2015) Review: High dose folic acid supplementation in women with epilepsy: Are we sure it is safe?'. Seizure: European Journal Of Epilepsy 27: 51-53
10. Harden C, Pennell P, Koppel B, Hovinga C, Gidal B, Meador K, Hopp J, Ting T, Hauser W, Thurman D, Kaplan P, Robinson J, French J, Wiebe S, Wilner A, Vazquez B, Holmes L, Krumholz A, Finnell R, Shafer P and Le Guen C n.d., Management issues for women with epilepsy-Focus on pregnancy (an evidence-based review): III. Vitamin K, folic acid, blood levels, and breast-feeding. Epilepsia 50(5): 1247-1255
National Guidelines
Epilepsies in children, young people and adults NICE guideline [NG217] NICE Published: 27 April 2022
Royal College of Obstetricians and Gynaecologists (2016) Epilepsy in Pregnancy. Green-top Guideline No. 68. Royal College of Obstetricians and Gynaecologists
SIGN guidelines (2015) Diagnosis and management of epilepsy in adults
Author: Morley K (2023) Preparing for pregnancy. Available from: www.womenwithepilepsy.co.uk
This website was founded by Kim Morley, an epilepsy specialist midwife practitioner in 2005. The process of rebuilding the website was started in June 2016 to benefit as many women with epilepsy as possible and the professionals who care for them.
For the latest news click on: Professional page Visitor counter added 19/08/16 & reached 100,000 08/2022. Reset 9/22